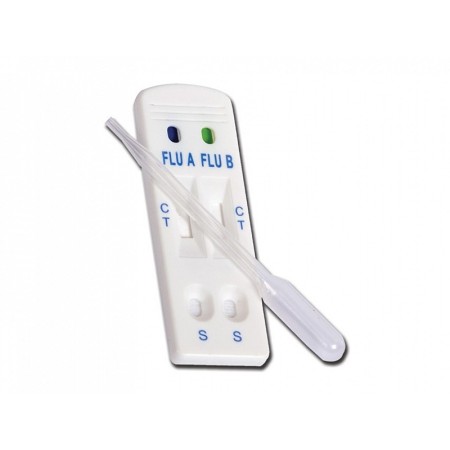
Influenza A/B test - pack 30 pcs.
The in vitro test for influenza A/B can detect type A and B virus antigens (nucleoproteins) taken from respiratory samples. The test is designed to detect the various subtypes of influenza A, (H1N1, H3N2, H5N1, H5N3, H7N3, H9N2).
It is performed through the use of nasal swabs that allow the necessary respiratory samples to be taken. User-friendly with results in 10-15 minutes. Include device for rapid test, swab, tube, reagent.
For professional use only.
WARNING: SHORT-TERM...
It is performed through the use of nasal swabs that allow the necessary respiratory samples to be taken. User-friendly with results in 10-15 minutes. Include device for rapid test, swab, tube, reagent.
For professional use only.
WARNING: SHORT-TERM...
Secure payments
by credit card, Paypal and bank transfer.
by credit card, Paypal and bank transfer.
Fast shipping
with Poste, DPD, FedEx, DHL and UPS couriers
with Poste, DPD, FedEx, DHL and UPS couriers
Customer service
via Whatsapp + 39.371.43.61.201
via Whatsapp + 39.371.43.61.201
Description
The in vitro test for influenza A/B can detect type A and B virus antigens (nucleoproteins) taken from respiratory samples. The test is designed to detect the various subtypes of influenza A, (H1N1, H3N2, H5N1, H5N3, H7N3, H9N2).
It is performed through the use of nasal swabs that allow the necessary respiratory samples to be taken. User-friendly with results in 10-15 minutes. Include device for rapid test, swab, tube, reagent.
For professional use only.
WARNING: SHORT-TERM TESTS.
The tests have a duration of 24 months from the date of manufacture. The stock could range from 10 to 22 months.
Specifications:
- BD/RDM: 1704450
- NDT: W0105099004
- Paraf: 977810694
- EAN13: 8023279245370
It is performed through the use of nasal swabs that allow the necessary respiratory samples to be taken. User-friendly with results in 10-15 minutes. Include device for rapid test, swab, tube, reagent.
For professional use only.
WARNING: SHORT-TERM TESTS.
The tests have a duration of 24 months from the date of manufacture. The stock could range from 10 to 22 months.
Specifications:
- BD/RDM: 1704450
- NDT: W0105099004
- Paraf: 977810694
- EAN13: 8023279245370
Data sheet
6 other products in the same category: